Venkatasai is a leading innovator in the field of pharmaceutical reference standards, offering high-quality Reference Standards of Abacavir, including both pharmacopeial and non-pharmacopeial impurities, metabolites, stable isotope-labeled compounds, and nitrosamines (N-NO products). Our Abacavir impurity reference standards play a vital role in pharmaceutical research, supporting product development, ANDA and DMF filings, quality control (QC), method validation, and stability studies. They are also instrumental in identifying unknown impurities and evaluating genotoxic potential. All Abacavir-related products are thoroughly characterized and supplied with comprehensive Certificates of Analysis (COA) and analytical data that comply with regulatory standards. EP/USP traceable standards can also be provided upon request. Furthermore, all supplied products undergo periodic re-testing to ensure continued quality and reliability.
X
Venkat Sai Life Science
x
- Upcoming Expo
Cafedrine Related Products
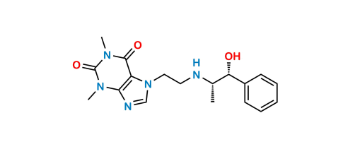
CAT No: VS-C232000
CAS No: 58166-83-9
Mol.F.: C18H23N5O3
Mol.Wt.: 357.4
Status: Custom Synthesis
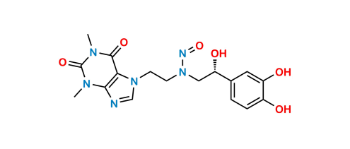
CAT No: VS-C232002
CAS No: NA
Mol.F.: C17H20N6O6
Mol.Wt.: 404.4
Status: Custom Synthesis
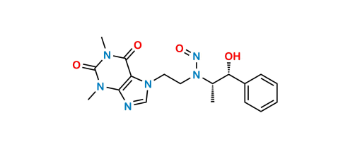
CAT No: VS-C232001
CAS No: NA
Mol.F.: C18H22N6O4
Mol.Wt.: 386.4
Status: Custom Synthesis